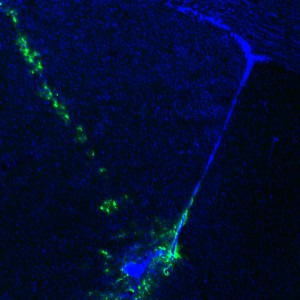
Adenoviral targeting of the ventral tip of the subventricular zone. GFP labeling delineates the site of injection and the injection track.
Access workflows from our group on Github.
Our laboratory employs a variety of in vitro and in vivo techniques to study the consequences of morphogenic and mitogenic pathway activation in the stem cell niche. Through ongoing collaborations with neurosurgeons and other researchers, we work constantly to translate our findings in research models to studies of human brain specimens. Most recently, through collaborative work with the Irish Lab @ Vanderbilt, we have developed dissociation and staining techniques which allow measurement of 35+ phospho/protein epitopes per cell using mass cytometry, enabling us to measure signaling pathway activity at the single-cell level in millions of cells per specimen. The laboratory is now applying these methods to map signaling and drug response in many grades of brain tumors.
The use of genetically modified mouse models is also a primary focus. Our expertise in stereotaxic delivery of replication-incompetent virus allows us to genetically manipulate progenitor cells of interest and follow their progeny over time. These techniques have been adapted to allow labeling of populations of stem cells at specific developmental ages and even at specific sites within the stem cell niche, and can also be exploited to study potential cells of origin in mouse models of brain tumor development.
In addition to in vivo work, the lab also specializes in confocal microscopy, neural stem cell culture methods and 3D visualization of the neural stem cell niche through en-face dissection of the region of interest. Immunohistological staining and mRNA expression analysis are also applied extensively to study both mouse and human tissue.
