“Minimizing the Reorganization Energy of Cobalt Redox Mediators Maximizes Charge Transfer Rates from Quantum Dots” selected as VINSE Spotlight Publication
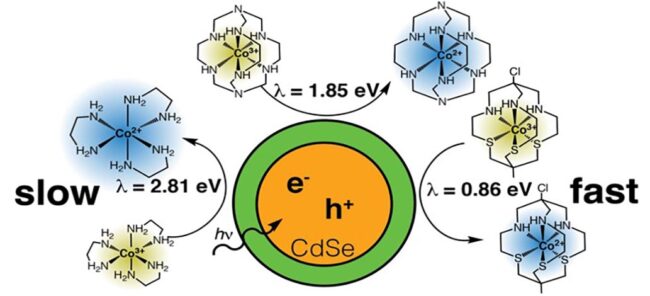
About the author: Madeleine Fort is working on her PhD in Interdisciplinary Materials Science under the guidance of Dr. Janet Macdonald. In this recently published article, she, and Sophia Click from Dr. Sandra Rosenthal’s lab, observed the effect of the reorganization energy of a charge acceptor on electron transfer rates from cadmium-based quantum dots. This was done by using steady state fluorimetry and time-resolved photoluminescence to monitor the fluorescence of the quantum dots with the addition of several cobalt(III) complexes whose reorganization energies varied over 2 eV. Charge transfer to the lower reorganization energy complexes was 2 orders of magnitude faster than the higher energy complexes, achieving sub-nanosecond lifetimes. This is a new path towards increasing the efficiency a myriad of technologies from dye-sensitized solar cells to the production of hydrogen from water splitting.